WHAT ARE HARMFUL ALGAE?
Phytoplankton blooms, micro-algal blooms, toxic algae, red tides, or harmful algae, are all terms for naturally occurring phenomena. About 300 species of micro algae are reported at times to form mass occurrence, so called blooms. Nearly one fourth of these species are know to produce toxins. The scientific community refers to these events with a generic term, ‘Harmful Algal Bloom’ (HAB), recognising that, because a wide range of organisms is involved and some species have toxic effects at low cell densities, not all HABs are ‘algal’ and not all occur as ‘blooms’.

Table of Contents
Why are they harmful?
Harmful algae and marine food resources
Toxic effects on humans
Which are the causative organisms?
Harmful algae as invasive species
References of interest
Table of Contents
Why are they harmful?
Harmful algae and marine food resources
Toxic effects on humans
Which are the causative organisms?
Harmful algae as invasive species
References of interest
WHY ARE THEY HARMFUL?
Proliferations of microalgae in marine or brackish waters can cause massive fish kills, contaminate seafood with toxins, and alter ecosystems in ways that humans perceive as harmful. A broad classification of HABs distinguishes two groups of organisms: the toxin producers, which can contaminate seafood or kill fish, and the high-biomass producers, which can cause anoxia and indiscriminate kills of marine life after reaching dense concentrations. Some HABs have characteristics of both.

Olive green discolouration of the waters of False Bay by the dinoflagellate Gymnodinium cf. mikimotoi – In addition to faunal mortalities, this species is also the cause of a type of NSP and the production of aerosol toxins causing skin and respiratory irritations to humans. Photograph by G.C. Pitcher.
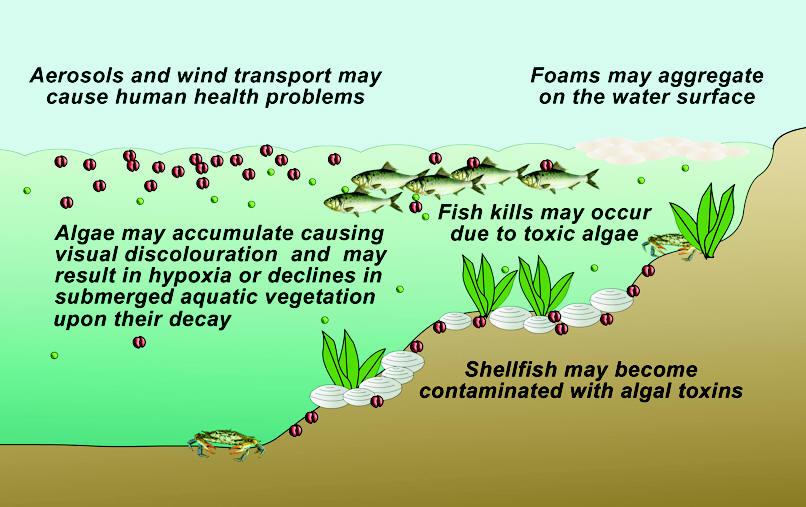
Although HABs occurred long before human activities began to transform coastal ecosystems, a survey of affected regions and of economic losses and human poisonings throughout the world demonstrates very well that there has been a dramatic increase in the impacts of HABs over the last few decades and that the HAB problem is now widespread, and serious. It must be remembered, however, that the harmful effects of HABs extend well beyond direct economic losses and impacts on human health. When HABs contaminate or destroy coastal resources, the livelihoods of local residents are threatened and the sustenance of human populations is compromised.
HARMFUL ALGAE AND MARINE FOOD RESOURCES
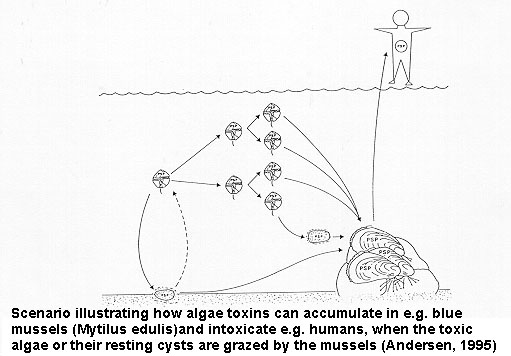
The impact of harmful microalgae is particularly evident when marine food resources,e.g. aquaculture, are affected. Shellfish and in some cases finfish are often not visibly affected by the algae, but accumulate the toxins in their organs. The toxins may subsequently be transmitted to humans and through consumption of contaminated seafood become a serious health threat. Although the chemical nature of the toxins is very different, they do not generally change or reduce significantly in amount upon cooking; neither do they generally influence the tate of the meat. Unfortunately, detection of contaminated seafood is not straight forward, and neither fishermen nor consumers can usually determine whether seafood products are safe for consumption. To reduce the risk of serious seafood poisoning intensive monitoring of the species composition of the phytoplankton is required in the harvesting areas in connection with bioassays and/or chemical analyses of the seafood products.

Hydrogen sulphide poisoning – In March 1994, a massive marine mortality was experienced in St Helena Bay on the South African west coast22. This event was caused by the entrapment and subsequent decay of an expansive red tide dominated by the dinoflagellates Ceratium furca and Prorocentrum micans. Marine life died from both suffocation and hydrogen sulphide poisoning. Oxygen concentrations were maintained at <5.5 ml.l-1 in the bottom waters of the Bay and hydrogen sulphide, subsequently generated by anaerobic bacteria in the absence of dissolved oxygen, was recorded in excess of 50 µmol.l-1. Approximately 60 tons of rock lobster and 1500 tons of fish, comprising about 50 species, washed ashore. The mullet Liza richardsoni made up the bulk of the fish mortality, with the remainder dominated by sharks and bottom-dwelling fish. The production of hydrogen sulphide gas caused the sea to turn black, and the event was soon dubbed the “black tide” by the media. Although common in the northern Benguela, this was the first recorded incidence of hydrogen sulphide poisoning in the southern Benguela. Photograph – The Argus
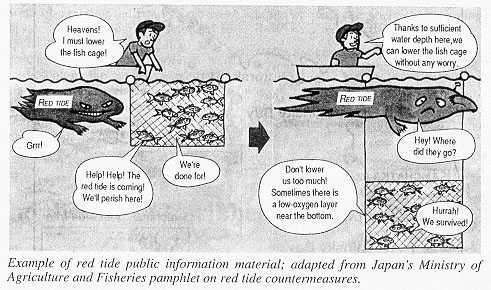
In addition to posing serious health risks to consumers of seafood, some microalgae may have devastating effects on fish and other marine life, both in wild and aquacultures. Several species of micro algae belonging in different taxonomic groups can produce toxins which damage fish gills by hemolytic effects. This has resulted in extensive fish kills with major economic losses. A comprehensive economic analysis of the global impact of harmful algal events on the aquaculture industry is not available, but the economic losses from single event in North America and especially Japan have on several occasions amounted to more than US$ 10 million. On one particular occasion the raphidophyte flagellate Chattonella antiqua killed US$ 500 million worth of caged fish in Japan. Additional losses may be inferred from public miscredit of seafood products due to misunderstandings and misinformations about harmful algal events.
In developing countries, seafood often constitutes an important or even sole source of food and protein, especially in costal areas. With the increasing problems of overfishing, aquaculture may become an increasingly important alternative for the supply of seafood. However, to minimize the risk of sea-food poisonings and the risk of major economic losses due to fish kills, it is important to establish adequate surveillance programmes and quality control of the seafood products which will often require expert assistance from countries which have longstanding experience in this matter.
TOXIC EFFECTS ON HUMANS
Particularly in the tropics people are often harassed by diseases and syndromes due to consumption of seafood contaminated by algal toxins. Some of these diseases may be fatal. There is currently no international record of the number of incidents of human intoxication caused by contaminated seafood. The numbers appearing in presentations at international meetings are undoubtedly underestimates, as many cases and even fatalities can be assumed to pass undiagnosed and hence unreported in the official reports.
Six human syndromes are presently recognized to be caused by consumption of contaminated seafood:
- amnesic shellfish poisoning – ASP
- ciguatera fish poisoning – CFP
- diarrhetic shellfish poisoning – DSP
- neurotoxic shellfish poisoning – NSP
- paralytic shellfish poisoning – PSP
- Azaspiracid shellfish poisoning- AZP
Other threats to human health are posed by blue-green algal toxins in drinking water which may cause severe damage or be tumor promoters.

Amnesic shellfish poisoning – ASP
This syndrome can be life-threatening. It is caused by domoic acid that accumulates in shellfish, but the disease can apparently also be fish borne, so the risk to humans may be more serious than previously believed. It is characterized by gastrointestinal and neurological disorders including loss of memory. Human ASP intoxication is presently known primarily from Canada, but the causative diatoms occur in many parts of the world, so considerable care should be exercised during blooms of species of the diatom Pseudo-nitzschia.
Ciguatera fish poisoning – CFP
This poisoning, transmitted by several tropical reef fish, is generally not lethal, although fatalities have been documented. Ciguatera produces gastrointestinal, neurological and cardiovascular disturbances, and recovery often takes months or even years. It is widely distributed in the tropics; thus in the period 1960-1984, there were a total of 24.000 cases of ciguatera in French Polynesia alone. Evidence is accumulating that disturbances of coral reefs by hurricanes, tourist activity etc. increase the risk of ciguatera by providing more suitable habitats for the benthic dinoflagellates (see causative organisms). There is at present no easy method to routinely measure the toxins (ciguatoxin and maitotoxin) that cause ciguatera fish poisoning.
Diarrhetic shellfish poisoning – DSP
This is a wide spread type of shellfish poisoning which causes gastrointestinal disturbances with diarrhea, vomiting, and abdominal cramps. It is not fatal, and the patients usually recover within a few days. There are thousands of reported incidents from developed countries, e.g. 5000 in Spain in 1981 alone, but with the pathological picture of DSP, many incidents may be regarded as an ordinary stomach disorder, and therefore remain unreported. Chronic exposure to DSP is suspected to promote tumor formation in the digestive system.
Neurotoxic shellfish poisoning – NSP
Until recently this syndrome has been restricted to the Gulf of Mexico, but in 1993 it was reported also from New Zealand. It is characterized by gastrointestinal and neurological disturbances usually with recovery within a few days. Toxic aerosols formed by wave action may cause asthma-like symptoms.
Paralytic shellfish poisoning – PSP
This is a life-threatening syndrome with neurological effects. There is no known antidote to PSP. The known global distribution has increased markedly over the last few decades. Each year about 2000 cases of PSP are reported with 15 % mortality.
Azaspiracid shellfish poisoning- AZP
This is a newly identified marine toxic syndrome, caused by consumption of mussels contaminated by the dinoflagellate. The typical symptoms of AZP resemble those of DSP, whereas neurotoxic symptoms are also observed in some poisoning incidents. The condition is not fatal, and full recovery usually occurs within 2-3 days of exposure. Azaspiracid has a global distribution and is tested for and regulated in Europe.
WHICH ARE THE CAUSATIVE ORGANISMS?
Many of the syndromes and other harmful effects pertain to occurrences of dinoflagellates. There is, however, an increasing number of species recognized as toxic in other algal classes, and harmful species are now found in at least 5 groups of algae:
Dinophyceae (= Dinoflagellates)
Species of Dinoflagellates are responsible for ciguatera, DSP, NSP, and PSP. Ciguatera is caused by Gamberidiscus toxicus, and perhaps also by some benthic species of the genera Ostreopsis and Prorocentrum. Most of these species can readily be identified by trained taxonomists.
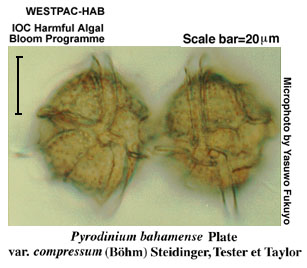
PSP is caused by several species. Most cases are caused by Pyrodinium bahamense var. compressum and species of Alexandrium. The latter genus comprises about 30 species which are difficult to identify. They are distinguished by small differences in the plate structure of the plates covering the cell.

DSP is caused by several species of Dinophysis and some benthic species of Prorocentrum. Dinophysis is a large genus with some 100 described species many of which pose considerable taxonomic problems. Species are distinguished only by morphological features such as size, shape, presence/absence of chloroplasts etc. Thus the circumscription of many species is vague and a comprehensive taxonomic revision of this genus is needed.
NSP is caused by Karenia Brevis (Gymnodinium breve).
In addition to the human syndromes caused by dinoflagellates, extensive fih kills have been caused by several so-called unarmoured species belonging in the genera Amphidinium, Cochlodinium, and Gymnodinium. Identification of these is generally difficult and requires examination of live cells.
Prymnesiophyceae (= Haptophycae)
Species of Chrysochromulina and especially Prymnesium have caused fish kills on many occasions, but have never been reported to be a threat to human health. The species of both genera are generally well circumscribed, but critical identification to species level requires electron microscopy to examine the micromorphology of the scales covering the cell surface.
Raphidophyceae
This class includes species which are responsible for the most extensive fish kills reported so far. Most species deteriorate upon preservation and critical identification generally requires observation of live cells.
Bacillariophyceae (= Diatoms)
ASP is caused by Pseudo-nitzschia australis, P. multiseries, and perhaps P. pseudodelicatissima. The two latter species are widely distributed in both the Northern and Southern Hemispheres, while the former occurs mostly in the Southern Hemisphere. Species identification is difficult often requiring electron microscopy, and misidentifications probably occur in the literature.
Cyanophyceae (= Blue-green algae)
In the marine environment, only few species of blue-green algae cause problems, e.g. species of Trichodesmium and Nodularia, and these have not been associated with human syndromes so far. A wide variety of blue-green algae cause serious problems in fresh and brackish water environments and may be a serious health problem where surface water is used for drinking water supplies.
WHAT TO DO ABOUT THEM
Identification of the causative species
When harmful algae occur, an immediate and essential action is reliable identification of the causative species involved for a first assessment of the potential risks. Many species are difficult to identify and advanced taxonomic training is necessary. Application of modern taxonomic concepts requires in some cases electron microscopy for critical identification, and this equipment is not generally available in many countries (particularly developing countries). There is often also a scarcity of scientific literature and/or limited access to libraries which is essential for both monitoring and scientific work.
Advice to those fishing, gathering shellfish or swimming
As it is impossible to see with the naked eye whether there are harmful algae in the water, examination of the water or seafood in the laboratory is necessary to determine the presence of harmful algae or algal toxins. Even after authorities have monitored an area there is no guarantee that there are no toxins present. The toxicity can vary from locality to locality and from day to day. Most authorities therefore monitor whether there is a potential danger of toxicity.
The best you can do is always to follow some general advice:
When fishing
- do not fish in the middle of an algal bloom/discoloured water
- never eat fish which are dead when caught
When gathering shellfish
- be aware of the intoxication symptoms
- if you experience any symptoms, keep a sample of the shellfish for toxicity testing
When swimming
- look for warnings against algal blooms
- never allow children to swim if there is a risk of algal toxins in the water
- never swim if you cannot see you feet when the water level is at your knees
See a doctor if you or your children get sick after eating seafood or swimming
To give the doctor the best background for finding out why you are sick it is adviseable to bring a sample of the seafood, or the water in which you swam.
WHO is currently preparing guidelines for safe recreational water environments. More information can be obtained from:
WHO, Water, Sanitation and Health Unit
Department of the Protection of the Human Environment
20 Avenue Appia,
CH-1211 Geneva 27
Tel. +41 22 791 35 37
Fax. +41 22 791 41 59
e-mail: bartramj@who.ch
HARMFUL ALGAE AS INVASIVE SPECIES
Several countries have prepared, or are in the process of preparing, white, grey and black lists of alien invasive species (Re e.g. European Strategy on Invasive Alien Species). With regard to marine species, harmful algae naturally come up as a group of organisms of interest. As the number of potential harmful invasive micro algae species is beyond what is practical to list, one approach may be to list those:
Criteria 1: Toxic
Subcriteria:
* Occurring in same climate zone or slightly warmer areas (re global warming)
* Cyst/resting stage forming species
If these criteria are applied to the IOC Taxonomic Reference List it may provide a reasonable first listing of potentially invasive HAB species in a given area/region. This is without considering if introduction would be with ballast, aquaculture, fouling etc. The listing of potentially toxic alien invasive HAB species may be of relevance for monitoring both in relation to the coming ballast water convention and for national or regional strategies on invasive species.
It is difficult to be sure that a given micro algae species has been introduced anywhere. On the other hand, it is quite common to hear that a new HAB phenomenon somewhere is caused by an invader. An expected introduction should be tested at sites that have been extensively and intensively monitored beforehand. Still, this may not be sufficient to prove an introduction.
For a more focused ranked risk assessment, bloom forming species are probably higher risk (for any vector) because the source propagule pressure will be higher. Another important issue is the time an organism can resist in e.g. ballast water. Species with resting stages (e.g., cysts or spores) would also pose a higher risk. However, non-spore-forming diatoms also can survive for long periods of time. Species with a history of being invasive in other parts of the world pose probably higher risk as well, however, most bloom-forming species that aren’t already considered cosmopolitan would still make the list—so that doesn’t really narrow it much (relative to the amount of work involved in ferreting out the history for each species). If you are focused on the ballast vector, coastal and estuarine species are higher risk as are anything with a broad salinity tolerance (under current ballast exchange requirements). If you have the time and energy, the better approach would be to map conditions in the receiving systems with the species tolerances/requirements. A species we consider low risk to a freshwater system might be high risk to a marine port or estuary and vice versa.
In thinking about the HAB species that should be on such a list do not leave out the freshwater cyanobacteria. One cyanobacterial HAB species that should be considered for the list is the cyanobacteria Cylindrospermopsis raciborskii. It is a relatively recent invader in many US lakes, including a recent appearance in western Lake Erie and some of the inland lakes in the Great Lakes region, but is also present in many locations (both tropical and more recently temperate) worldwide.
Australia has listed since 1989 Gymnodinium catenatum, Alexandrium tamarense/catenella, Alexandrium minutum for which strong circumstantial evidence exists (Bolch & de Salas, Harmful Algae 2007) that they had been introduced to Australia, i.e. further spreading via domestic ballast water needs to be prevented. Pfiesteria piscicida was on a list of future pests. This taxon has now been confirmed from Australian ballast water and Australian ports, but its introduced status is unclear. PARK et al. Applied Environmental Microbiology 2007.
In New Zealand there are no HAB species designated as Unwanted under New Zealand’s Biosecurity Act, although Biosecurity NZ has been involved in some risk profiling work which has included HABs. Regarding criteria to list potential invaders, climate zones often don’t reflect actual disctributions of introduced species/ species spread as they often spread to areas beyond the thermal ranges of their native/common distributions. Criteria need to include some indication of potential harm—harmful species are not necessary cyst forming.
REFERENCES OF INTEREST
Wyatt, T and J.T. Carlton. 2002. Phytoplankton introductions in European coastal waters: why are so few invasions reported? In: CIESM Workshop Monographs n°20: Alien marine organisms introduced by ships in the Mediterranean and Black seas – Istanbul (Turkey), 6-9 November 2002, 136 p. (268 ref.) http://www.ciesm.org/online/monographs/Istanbul.html
Dickman, M. and F. Zhang. 1999. Mid-ocean exchange of container vessel ballast water. 2: Effects of vessel type in the transport of diatoms and dinoflagellates from Manzanillo, Mexico to Hong Kong, China. Marine Ecology Progress Series 176: 253-262.
Hallegraeff, G.M. 1998. Transport of toxic dinoflagellates via ships’ ballast water: bioeconomic risk assessment and efficacy of possible ballast water management strategies. Marine Ecology Progress Series 168: 297-309.
Hallegraeff, G.M. and C.J. Bolch. 1991. Transport of toxic dinoflagellate cysts via ships’ ballast water. Marine Pollution Bulletin 22(1): 27-29.
Hallegraeff, G.M. and C.J. Bolch. 1992. Transport of dinoflagellate cysts in ships’ ballast water: implications for plankton biogeography and aquaculture. Journal of Plankton Research 14: 1067-1084.
Jones, M.M. 1991. Marine organisms transported in ballast water: a review of the Australian scientific position. Bureau of Rural Resources Bulletin No. 11, Australian Government Publishing Service, Canberra. 48 pp.
Lilly, E.L., D.M. Kulis, P. Gentien and D.M. Anderson. 2002. Paralytic shellfish poisoning toxins in France linked to human-introduced strain of Alexandrium catenella from the western Pacific: evidence from DNA and toxin analysis. Journal of Plankton Research 24(5): 443-452.
McCarthy, H.P. and L.B. Crowder. 2000. An overlooked scale of global transport: phytoplankton species richness in ships’ ballast water. Biological Invasions 2:321-322.
McCollin, T.A., J.P. Hamer and I.A.N. Lucas. 1999. Transport of phytoplankton via ship’s ballast into ports around England and Wales. In J. Pederson (ed.). Marine Bioinvasions: Proceedings of the First National Conference. Massachusetts Institute of Technology, Cambridge, Massachusetts: 282-288.
McMinn, A, G.M. Hallegraeff, P. Thomson, A.V. Jenkinson and H. Heijnis. 1997. Cyst and radionucleotide evidence for the recent introduction of the toxic dinoflagellate Gymnodinium catenatum into Tasmania waters. Marine Ecology Progress Series 161: 165-172.
Patil, J.G., R.M. Gunasekera, B.E. Deagle, N.J. Bax and S.L. Blackburn. 2005. Development and evaluation of a PCR based assay for detection of the toxic dinoflagellate, Gymnodinium catenatum (Graham) in ballast water and environmental samples. Biological Invasions 7(6): 983-994.
Zhang, F. and M. Dickman. 1999. Mid-ocean exchange of container vessel ballast water. 1: Seasonal factors affecting the transport of harmful diatoms and dinoflagellates. Marine Ecology Progress Series 176: 243-251.
Xiaoping Wu, Li Hou, Xucong Lin, Zenghong Xie,Chapter 12-Application of Novel Nanomaterials for Chemo- and Biosensing of Algal Toxins in Shellfish and Water,Editor(s): Xiaoru Wang, Xi Chen,In Micro and Nano Technologies,Novel Nanomaterials for Biomedical, Environmental and Energy Applications,Elsevier,2019,Pages 353-414.
